Contents
- A patently wrong regime: COVID Vaccine
A patently wrong regime: COVID Vaccine
Context:
- In 2020, India and South Africa submitted a joint petition to the World Trade Organization (WTO), requesting a temporary suspension of rules under the 1995 Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS). The request for waiver has, since, found support from more than 100 nations.
- But a small group of states — the U.S., the European Union, the U.K. and Canada among them — continues to block the move. Their reluctance comes despite these countries having already secured the majority of available vaccines, with the stocks that they hold far exceeding the amounts necessary to inoculate the whole of their populations.
- Intellectual property regime has acted as a lethal barrier to the right to access health. Even request for a temporary waiver is not getting accepted.
Relevance:
GS-III: Indian Economy (International Trade, Planning usage and Mobilisation of resources), Intellectual Property Rights
Mains Questions:
Over the last few decades, intellectual property rules have served as a lethal barrier to the right to access healthcare. Discuss in the light of distribution of life-saving drugs in the fight against COVID-19. (10 marks)
Dimensions of the Article:
- What is a Patent?
- Patent protection in India (Details are not so important)
- Blockades in making Vaccines Available everywhere
- One important waiver: Compulsory licensing
- Why should waiver be provided?
- Creating a patent pool – Way Forward
What is a Patent?
- A patent is an exclusive right granted for an invention, which is a product or a process that provides, in general, a new way of doing something, or offers a new technical solution to a problem.
- To get a patent, technical information about the invention must be disclosed to the public in a patent application.
- Patent is provided for the period of 20 years. Exclusive rights are only applicable in the country or region in which a patent has been filed and granted, in accordance with the law of that country or region. Patent rights are usually enforced in a court on the initiative of the right owner.
- In India, Patents are provided by Indian Patent Office. Any appeal against the order or violation goes to Intellectual Property Appellate Board.
Justification for patents:
- People have something of a natural and moral right to claim control over their inventions.
- Exclusive rights promote invention.
- Individuals should be allowed to benefit from their labour and merit.
- Innovation is an expensive activity to undertake.
However, these patents have been taken as a medium to own monopoly.
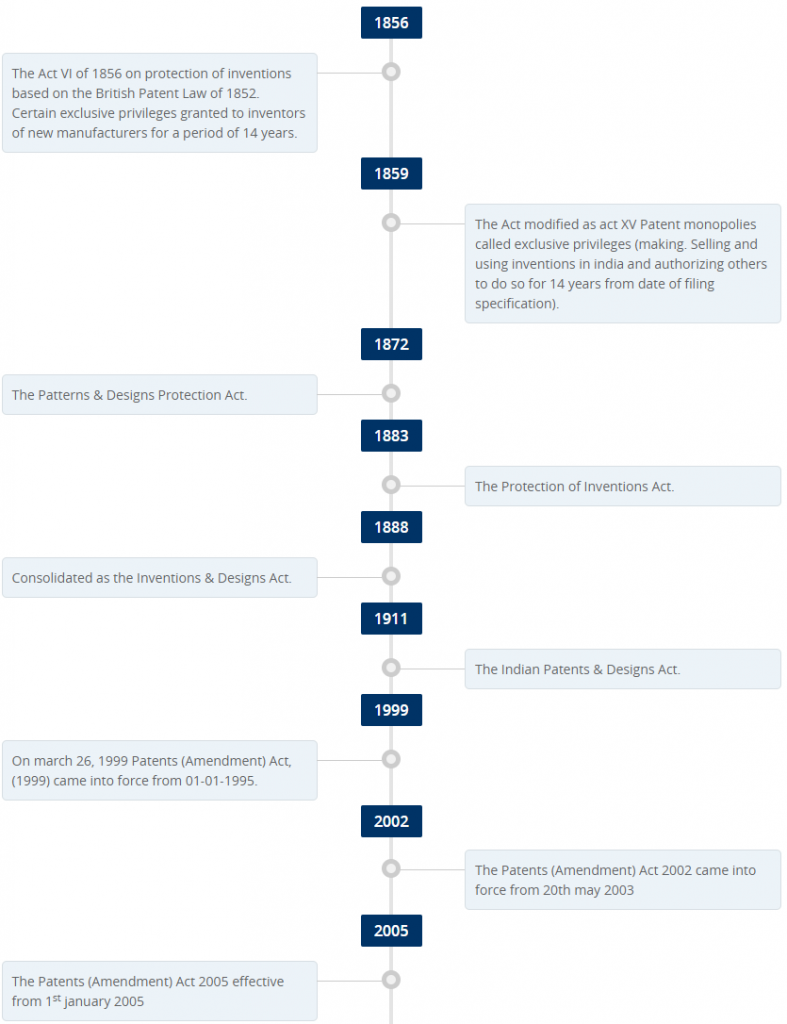
Patent protection in India (Details are not so important)
- The first legislation in India relating to patents was the Act VI of 1856 – But it was repealed by Act IX of 1857 because it had been enacted without the approval of the British Crown
- Fresh legislation for granting ‘exclusive privileges’ was introduced in 1 859 as Act XV of 1859.
- In 1872, the Act of 1859 was renamed as “The Patterns and Designs Protection Act” and it was consolidated to provide protection relating to designs.
- The Indian Patents and Designs Act, 1911, (Act II of 1911) replaced all the previous Acts. However, after Independence, it was felt that the Indian Patents & Designs Act, 1911 was not fulfilling its objective.
- Based on the recommendations of a Committee formed by the Indian Government after independence, the 1911 Act was amended in 1950.
- In 1952, an amendment was made to provide compulsory licence in relation to patents in respect of food and medicines, insecticide, germicide or fungicide and a process for producing substance or any invention relating to surgical or curative devices.
- In 1957, the Government of India appointed Justice N. Rajagopala Ayyangar Committee to examine the question of revision of the Patent Law and advise government accordingly and the committee recommended retention of the Patent System, despite its shortcomings. In 1959, the committee objected this regime on ethical grounds.
- India adopted a new Patents Act in 1970 and this led to the rise in generic medicines production in India. The Patents Act of 1970 has Amended a number of times. A new Patent Rules, 2003 was introduced (by replacing the earlier Patents Rules, 1972) since, under the Patents Act 1970 the Central Government is empowered to make rules for implementing the Act and regulating patent administration.
India and Generic medicines
- India is now the largest generic medicine producer in the world. India’s efforts on HIV and Cancer drugs are well appreciated.
- A generic drug is a medication created to be the same as an already marketed brand-name drug in dosage form, safety, strength, route of administration, quality, performance characteristics, and intended use.
- These similarities help to demonstrate bioequivalence, which means that a generic medicine works in the same way and provides the same clinical benefit as the brand-name medicine. In other words, you can take a generic medicine as an equal substitute for its brand-name counterpart.
Blockades in making Vaccines Available everywhere
- Even when approval for marketing of a vaccine/drug is granted, it will be impossible for it to be made instantly available across the world.
- This is because even after approval for commercial production is granted, say, in one country, in order for the product to be available to the rest of the world, approvals will be required in each and every country.
- Then countries will have to gear up for instant manufacturing and marketing of the drug.
- For this to happen, continuous dialogue has to take place among innovators, manufacturers and supply chains.
- This requires massive efforts by private players, governments and international organisations.
- Innovations may be the subject matter of patent applications around the world.
Will patents create roadblocks or is there a solution?
- Governments and international organisations need to arrive at a consensus in advance to ensure that the system is ready.
- Creating hindrances through exclusivity claims, in the wake of a pandemic, will result in dividing countries, corporations and international organisations.
- Under the TRIPS (Trade-Related Aspects of Intellectual Property Rights) regime, there are several tools such as compulsory licensing that are available to ensure access to medicines.
One important waiver: Compulsory licensing
For patents: when the authorities license companies or individuals other than the patent owner to use the rights of the patent — to make, use, sell or import a product under patent (i.e., a patented product or a product made by a patented process) — without the permission of the patent owner. Allowed under the WTO’s TRIPS (intellectual property) Agreement provided certain procedures and conditions are fulfilled.
The following conditions should be fulfilled by the applicant:
- Reasonable requirements of the public with respect to the patented invention have not been satisfied.
- Patented invention is not available to the public at a reasonably affordable price.
- Patented invention is not used in India.
Countries would be able to facilitate a free exchange of know-how and technology surrounding the production of vaccines.
Why should waiver be provided?
- Most of the general medicines are produced under the research of Government funding mechanism or donations. For example: 97% of the funding towards the development of the Oxford/AstraZeneca vaccine recently was public money. Then how can they create monopoly on this.
- The cost of innovation or R&D could be easily provided through a one-time payment or prize money rather a long-term patent.
Creating a patent pool – Way Forward
- One method by which aggregation and dissemination of innovative products can be ensured is by creating a patent pool.
- Patent pools are usually effective in aggregating, administering and licensing patents related to specific areas of technology.
- Such pools are usually managed by a central agency and the patents which become part of the pool are readily made available for licensing.
- All countries ought to have the right to implement these innovations without further permission from the patent-holders and without resorting to provisions such as compulsory licensing, state acquisition, etc.’
- Creation of a pool and immediate licensing will ensure that there are hundreds of manufacturers across the world. As a result, vaccines and medicines will be quickly available.
- Pooling of patent resources is also in line with the Doha Declaration on Public Health which is a part of the TRIPS agreement. This declaration recognises the need for taking measures to ‘protect public health’ and ‘promote access to medicines’.
- Public-private partnerships (PPP) need to be scaled up. Creation of the ‘PPP-pandemic patent pool’ at a global level, to pool all innovations, is the way forward.
-Source: The Hindu



