Content
- GLP-1 Drugs Use, Risks, and Regulation
- India’s Seafood Exports: From Growth to Global Competitiveness
GLP-1 Drugs Use, Risks, and Regulation
Why in News?
- DCGI (2026) intensified surveillance against unauthorised GLP-1 sales, targeting online pharmacies, clinics, wholesalers, amid rising misuse for cosmetic weight loss without proper prescriptions.
- 49 entities inspected nationwide, revealing violations like OTC sales, improper prescriptions, misleading advertisements, prompting warnings, notices, and potential license cancellation and legal action.
Relevance
- GS II (Governance & Social Justice)
- Regulation of pharmaceuticals (Drugs & Cosmetics Act, CDSCO, DCGI)
- Ethical issues in healthcare (misleading ads, influencer marketing, access inequity)
- Public health governance (NPCDCS, rational drug use)
- GS III (Science & Tech / Economy)
- Biotechnology & pharmacology (incretin-based therapies)
- Health economy (high-cost therapies, pharma market expansion)
- Digital governance (e-pharmacy regulation, AI pharmacovigilance)
Practice Question
- “The rise of GLP-1 drugs highlights both innovation in metabolic disease management and emerging regulatory challenges.”Discuss the governance, ethical, and public health implications of their increasing use in India. (250 words)
Static Background
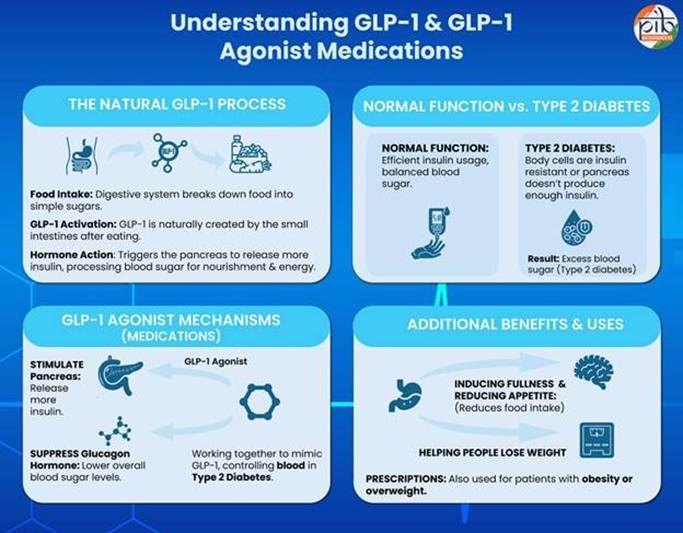
Diabetes & Hormonal Regulation
- Insulin lowers blood glucose by enabling cellular uptake, while glucagon increases glucose via hepatic release; both maintain homeostatic glucose balance in healthy individuals.
- Type 2 diabetes involves insulin resistance and relative insulin deficiency, combined with excess glucagon activity, leading to persistent hyperglycaemia and metabolic dysfunction.
GLP-1 Hormone (Incretin System)
- GLP-1 (incretin hormone) is secreted from intestines post-meal, enhancing glucose-dependent insulin secretion and suppressing glucagon release, ensuring efficient glucose regulation.
- It also delays gastric emptying and induces satiety, thereby reducing food intake and postprandial glucose spikes, linking metabolism with appetite regulation.
What are GLP-1 Drugs?
Definition
- GLP-1 receptor agonists are pharmacological agents that mimic incretin hormones, correcting dual defects of insulin deficiency and glucagon excess in type 2 diabetes patients.
Mechanism of Action
- These drugs increase insulin secretion and suppress glucagon, while delaying gastric emptying, leading to improved glycaemic control and sustained weight reduction.
- Appetite suppression via central satiety pathways reduces caloric intake, making them effective for obesity management alongside diabetes treatment.
Examples
- Common drugs include Semaglutide, Liraglutide, Tirzepatide (dual GIP+GLP-1), Dulaglutide, Exenatide, administered mainly via injectable pens or oral tablets (semaglutide).
Governance / Regulatory Framework
Legal & Institutional Setup
- Regulated under Drugs and Cosmetics Act, 1940, with CDSCO (under DCGI) as the apex authority ensuring drug approval, safety, quality, and ethical marketing practices.
Recent Actions (2026)
- March 2026 advisory directed manufacturers to stop misleading advertisements and non-prescription promotion, addressing rising misuse of GLP-1 drugs for non-therapeutic purposes.
- Nationwide inspections targeted e-pharmacies, wholesalers, wellness clinics, identifying violations like unauthorised sale and improper prescription practices, strengthening regulatory enforcement.
Prescription Norms
- GLP-1 drugs are Schedule H prescription drugs, permitted only through qualified specialists (endocrinologists, physicians, cardiologists) to ensure safe and supervised clinical use.
Economic Dimension
- India has ~101 million diabetics (ICMR-INDIAB 2023), making it a global hotspot, with rising obesity accelerating demand for advanced but expensive therapies like GLP-1 drugs.
- Monthly treatment costs of ₹8,000–₹15,000 limit access, creating health inequity, where affluent urban populations benefit disproportionately compared to vulnerable groups.
- Global GLP-1 market projected to exceed $100 billion by 2030, indicating massive pharmaceutical growth potential and commercialisation of metabolic disease treatment.
Social / Ethical Dimension
- Increasing off-label use for cosmetic weight loss reflects a shift from medical necessity to lifestyle consumption, driven by celebrity endorsements and social media trends.
- Ethical concerns include self-medication, body image pressure, and diversion of essential drugs, potentially depriving genuine diabetic patients of critical treatment access.
Health Risks and Safety Concerns
Common Side Effects
- Frequently reported effects include nausea, vomiting, dizziness, and gastrointestinal discomfort, especially during initial phases of therapy due to delayed gastric emptying mechanisms.
Severe Complications
- Serious risks include pancreatitis, kidney injury, bowel obstruction, and possible medullary thyroid cancer, necessitating strict medical supervision and patient selection criteria.
Clinical Concerns
- Long-term safety remains under evaluation, with contraindications in thyroid cancer history, severe gastrointestinal disorders, and caution required in multi-morbidity patients.
Governance Challenges / Criticisms
Regulatory Gaps
- Weak enforcement in e-pharmacy platforms and wellness clinics enables OTC misuse, undermining prescription norms and increasing risks of unsupervised consumption.
Supply Chain Issues
- Rising demand has led to black marketing, hoarding, and diversion, especially for high-demand drugs like semaglutide, affecting availability for genuine patients.
Ethical Marketing Issues
- Aggressive pharmaceutical promotion and influencer-driven advertising create information asymmetry, encouraging irrational demand and undermining evidence-based medical practices.
Capacity Constraints
- India faces shortage of endocrinologists (~1 per lakh population approx.), limiting specialist access and increasing dependence on unregulated or informal healthcare channels.
Environment / Technology Dimension
- Use of injectable pens generates biomedical waste, raising concerns about safe disposal and environmental sustainability in expanding chronic therapy markets.
- Digital platforms enable unauthorised online drug sales, but also offer solutions like e-prescriptions, AI-based pharmacovigilance, and real-time regulatory monitoring systems.
Way Forward
Strengthen Regulation
- Implement mandatory e-prescription verification systems and integrate e-pharmacies under CDSCO oversight, ensuring traceability and preventing unauthorised drug access.
Ethical Governance
- Strict enforcement of Drugs and Magic Remedies Act, 1954 to curb misleading advertisements, along with regulation of celebrity and influencer endorsements in healthcare marketing.
Public Health Approach
- Prioritise lifestyle interventions (diet, exercise) under programmes like NPCDCS, reducing over-reliance on pharmacological solutions for preventable metabolic disorders.
Improve Access & Equity
- Consider price regulation via NPPA and insurance inclusion, ensuring affordability and equitable access to GLP-1 drugs for clinically indicated patients.
Surveillance & Research
- Establish national registries and AI-driven pharmacovigilance systems to monitor long-term safety, adverse effects, and real-world outcomes of GLP-1 drug usage.
Prelims Pointers
- GLP-1 drugs mimic incretin hormone, not insulin, enhancing glucose-dependent insulin secretion and suppressing glucagon.
- Tirzepatide acts as a dual GIP + GLP-1 receptor agonist, offering enhanced metabolic effects.
- Regulatory authority: CDSCO under DCGI ensures approval, monitoring, and enforcement of drug standards in India.
- Type 1 diabetes involves insulin deficiency, while Type 2 diabetes involves insulin resistance and glucagon excess.
- BMI threshold in India: obesity defined as ≥25 kg/m², lower than global standards due to higher metabolic risk.
India’s Seafood Exports: From Growth to Global Competitiveness
Why in News?
- India’s seafood exports reached ₹62,408 crore (2024–25), registering sustained ~7% CAGR over 11 years, with frozen shrimp contributing ₹43,334 crore, indicating strong export concentration.
- India secured US MMPA comparability finding (2025), ensuring continued access to largest market (36.42% share) beyond deadline, alongside ₹39,272 crore public investment since 2015.
Relevance
- GS III (Economy)
- Agri-exports & Blue Economy
- Export diversification and value addition
- External trade vulnerability (market concentration, shrimp dominance)
- GS II (Governance)
- Policy interventions (PMMSY, digital traceability, SPS compliance)
- Trade diplomacy (US MMPA compliance, global market access)
Practice Question
- “India’s seafood exports have grown significantly, but sustainability and diversification remain key challenges.”Critically analyse the structural issues in India’s fisheries export sector and suggest measures for achieving global competitiveness. (250 words)
Static Background
Fisheries Sector Overview
- Fisheries sector supports ~30 million primary livelihoods and nearly 60 million across value chain, contributing to employment, food security, and rural income diversification.
- India is 2nd largest aquaculture producer globally, contributing ~8% of global fish production, driven by rapid expansion of brackish-water aquaculture systems.
Growth Trends
- Fish production increased from 141.64 lakh tonnes (2019–20) to 197.75 lakh tonnes (2024–25), reflecting consistent ~7% annual growth and sectoral commercialisation.
- Marine exports more than doubled from ₹30,213 crore (2013–14) to ₹62,408 crore (2024–25), indicating rising global integration and export competitiveness.
What are India’s Seafood Exports?
Export Composition
- India exports 350+ seafood varieties to ~130 countries, including frozen shrimp, fish, squid, cuttlefish, surimi products, dried and live seafood, reflecting increasing diversification.
- Share of value-added products increased from 2.5% to 11%, generating USD 742 million, indicating gradual shift from raw exports to processed, high-value products.
Major Markets
- USA accounts for 36.42% of exports, followed by China, EU, ASEAN, Japan, Middle East, highlighting dependence on few key markets with concentrated demand patterns.
Governance / Regulatory Framework (India)
Key Schemes & Interventions
- PMMSY drives sector transformation through quality seed production, aquaculture expansion, disease management, traceability systems, and capacity building across value chain.
- Promotion of high-value species (tuna, seabass, cobia, GIFT tilapia, tiger shrimp, seaweed) aims to diversify exports and access premium global markets.
Sustainability & Compliance Measures
- Deployment of Turtle Excluder Devices (TEDs) reduces by-catch, ensuring compliance with international conservation norms and export regulations like MMPA.
- National digital traceability framework enables end-to-end tracking, ensuring food safety, transparency, and compliance with SPS standards in US/EU markets.
Ease of Doing Business Reforms
- SIP digitisation via National Single Window System reduced approval time from 30 days to 72 hours, improving trade facilitation and efficiency.
- Waiver of SIP for SPF broodstock, fish oil, R&D samples, re-export inputs enhances ease of business and value-added processing capacity.
Economic Dimension
- Seafood exports contribute significantly to foreign exchange earnings and agri-export diversification, strengthening India’s blue economy strategy.
- Dominance of shrimp exports (₹43,334 crore) creates high commodity concentration risk, exposing sector to price volatility and disease outbreaks.
- Shift toward value-added processing (11% share) improves unit value realisation and global competitiveness, reducing reliance on bulk exports.
Social / Ethical Dimension
- Fisheries sector enables inclusive growth, supporting small-scale fishers, coastal communities, women workers, and marginal farmers under PMMSY interventions.
- Ethical supply chains through traceability and certification align with global standards on labour rights, sustainability, and responsible sourcing.
- However, export-driven mechanisation risks marginalising traditional artisanal fishers, creating intra-sectoral inequality and livelihood vulnerabilities.
Health & Quality Dimension
- Strengthened Sanitary and Phytosanitary (SPS) measures ensure compliance with stringent food safety norms in US and EU markets, protecting export credibility.
- Digital traceability allows rapid recall and quality monitoring, reducing risks of contamination, rejection, and reputational loss in global markets.
Environment / Sustainability Dimension
- Adoption of TEDs and EEZ sustainable fishing rules reduces by-catch and protects marine biodiversity and endangered species like sea turtles.
- Expansion of aquaculture must address risks of coastal ecosystem degradation, water pollution, and disease spread, requiring ecosystem-based management approaches.
Governance Challenges / Criticisms
Market Dependence
- Heavy reliance on shrimp (~major share) and US market (36.42%) creates vulnerability to trade barriers, regulatory shocks, and demand fluctuations.
Sustainability Gaps
- Incomplete adoption of TEDs and by-catch mitigation technologies undermines compliance with global environmental standards and export regulations.
Infrastructure Deficits
- Limited cold-chain infrastructure, modern harbours, inland logistics hubs restrict expansion of value-added exports and freshwater seafood supply chains.
Capacity Constraints
- Small-scale aquaculture faces challenges in technology adoption, disease control, and certification compliance, affecting productivity and export quality consistency.
Technology Dimension
- Digital traceability systems enhance transparency, compliance, and efficiency, enabling India to meet global food safety and sustainability benchmarks.
- Adoption of advanced hatchery technologies, cold-chain logistics, and processing units reduces post-harvest losses and improves export competitiveness.
Way Forward
Diversification Strategy
- Expand exports of high-value species (tuna, seabass, grouper, seaweed) to reduce dependence on shrimp and enhance resilience of export basket.
Value Addition Push
- Increase share of processed and ready-to-eat seafood beyond 11%, improving export margins and integration into global value chains.
Market Expansion
- Diversify towards UK, EU, ASEAN, West Asia markets, reducing vulnerability arising from US-centric export dependence.
Infrastructure Development
- Invest in integrated cold chains, inland export hubs, modern fishing harbours, ensuring efficient logistics and reduced post-harvest losses.
Sustainability Governance
- Ensure full compliance with MMPA, TED deployment, and EEZ regulations, aligning with SDG-14 (Life Below Water) commitments.
Policy Integration
- Integrate PMMSY with skilling, digital governance, and innovation ecosystems, balancing quantity growth with quality, sustainability, and competitiveness.
Prelims Pointers
- India contributes ~8% of global fish production and is 2nd largest aquaculture producer globally.
- Seafood exports reached ₹62,408 crore (2024–25), with shrimp contributing ₹43,334 crore.
- USA share: 36.42% of total seafood export value.
- PMMSY focuses on infrastructure, diversification, and export promotion.
- MMPA (USA) requires reduction of marine mammal by-catch for export eligibility.
- Fish production reached 197.75 lakh tonnes (2024–25) with ~7% annual growth.
- Value-added products share increased to 11% (USD 742 million).



