Contents:
- The race to find a cure for COVID-19
- Services at our doorstep: Delivery Workers are the new Front-line Force
- Ironing out wrinkles in India’s pandemic response
- Death by fireworks: Violations of rules in hazardous industries
THE RACE TO FIND A CURE FOR COVID-19
Focus: GS-III Science and Technology
Why in news?
- While efforts to develop an anti-COVID-19 vaccine are encouraging, safety issues are paramount.
- The epicentre of the pandemic has shifted from Wuhan district in China to the highly developed Western world.
- With no specific therapy or vaccine available at present, it is imperative that we fast-forward the development of efficacious vaccines and drug therapies against COVID-19.
- However, it must be emphasised that because vaccines are given to large populations, safety issues are paramount.
- The world is dealing with an unprecedented and unimaginably serious crisis.
- Therefore, the speed of vaccine development is crucial.
Encouraging developments
- The race for developing an anti-COVID-19 vaccine has begun.
- Reasonable scientific rationale and the information needed for vaccine development are available to all stakeholders in academia and industry.
- A large number of candidate vaccines based on different vaccine platforms, including delivering the virus genetic materials (RNA, DNA) or using synthetic biology to produce key viral proteins, have already been developed.
- Although based on some earlier experience of vaccines of other coronaviruses such as SARS and MERS, the speed with which the experimental vaccine has entered safety trials is unprecedented.
- The Serum Institute of India has also recently announced its readiness to start safety trials following animal experiments.
- According to a World Health Organization (WHO) report, more than 20 vaccine candidates are in advanced stages of development and will be ready for Phase-I safety trials.
- While these developments are encouraging, several questions will need to be answered for these candidates to move further.
Key Questions to be Addressed
- Although it is quite evident that humans mount a strong immune response and clear the viral load, the nature of the immune response and how to trigger it safely through vaccination will be key questions to address.
- How long the acquired immunity in humans will last is another important question to be asked before experimental vaccines move forward. We will need to know this because if the immunity is transient, then humans will be susceptible to reinfections.
- Before moving to Phase-II trials in a large number of healthy volunteers, we also have to ensure that the immune response induced by vaccination does not lead to any disease enhancement, as has been observed in cases of some experimental vaccines against the dengue virus, and in animal studies with an experimental vaccine against the SARS virus.
What is Urgently Needed?
- It is absolutely imperative that anti-COVID vaccines are developed as fast as possible and by pushing all reasonable vaccine candidates through animal and human clinical trials.
- However, it is also clear that it will not be possible to roll-out any efficacious vaccine for at least another year. An all-out effort to develop a vaccine against COVID-19 must continue if we have to stop its spread.
- With COVID-19 playing havoc across the world, therapeutic interventions, not only for curing severe cases of the disease but also for protecting all front-line healthcare workers, are urgently needed.
- Since developing new drugs is a complex and lengthy process, scientists and pharmaceutical companies have rushed to investigate and use drugs that have already been approved by regulatory authorities.
- Using available molecular and structural biology information on the virus, a group of scientists have analysed all interactions of the viral proteins with human proteins that are crucial for the virus to enter human cells and use the host cell machinery to rapidly reproduce itself.
- Of the nearly 70 short-listed molecules that may interrupt these key interactions, 24 happen to be already approved drugs which can now be tested in laboratory animal models as well as humans.
- However, the re-purposing of several drugs, alone or in combinations to treat COVID-19 patients, have already been reported.
- There are many success stories of curing patients of COVID-19 doing the rounds in different parts of the world, but these have managed to create more confusion than hope.
- Without any appropriate controls, careful dosing and safety concerns, such small experiments can only do more harm than good.
Controlled randomised trials
- Given the urgency of finding a cure, it is absolutely necessary to find out unequivocally what works well and what does not.
- For that conducting carefully controlled randomised trials is the only way to go. In a welcome move, the WHO has announced clinical trials called the ‘Solidarity Project’.
- Under this four drugs or drug combinations will be tested in many countries around the world.
- These candidates include the anti-Ebola drug, Remdesivir, Chloroquine, anti-HIV drugs, and the Ritonavir/Lopinavir combination, with or without Interferon-beta.
- Many countries have already signed up for these trials and all drug companies, including CIPLA from India, have agreed to supply sufficient quantity of drugs needed.
Conclusion
- Quickly getting all the stakeholders together is laudable and underscores the notion that everyone needs to fight the deadly virus together.
- Hopefully, these trials will lead to tangible drug therapies against COVID-19.
- It is most heartening to see scientists in academia and industrial partners coming together to fight a monumental public health crisis.
- The battle between pathogens and humans will continue but let us hope that we win the present one sooner than later.
SERVICES AT OUR DOORSTEP: DELIVERY WORKERS ARE THE NEW FRONT-LINE FORCE
Focus: GS-III Indian Economy
Why in news?
The ‘ferrymen’ of Wuhan, who have been providing groceries, food and medicines to citizens isolated in their homes, have been the backbone of cities that have been most hit by the novel coronavirus.
Delivery infrastructure or digitally enabled micro-logistics are key to how citizens navigate their ‘social distanced’ lives during the pandemic.
Delivery technologies are providing a key form of infrastructure that can be mobilised at short notice, dynamically and at scale.
Threat faced by delivery personnel in India
- Major delivery players in India have responded by publicising consumer awareness, restricting dynamic pricing (Amazon and Flipkart) and contact-less delivery protocols.
- Ola offers ₹1,000 a day for lost income for up to 21 days for drivers or their family members who contract the virus.
- Flipkart is reportedly mapping impacted areas to ensure that delivery agents are aware and least affected as they continue to offer relief to elite and middle-class households who have been the first-wave of likely infected citizens.
- However, company follow-through is in question.
- Reports show that delivery platforms have not yet implemented some of these protocols.
- Indian mobility platforms have not yet offered to extend or waive loans and rental fees for lease drivers.
Facing risks
- It is only when such infrastructure breaks or is in decay that public concern around it grows.
- The COVID-19 pandemic exacerbates the risk faced by delivery agents.
- They are unable to take a break from work, work from home, or access the superfluous resources that white-collar and professionals in other fields are deriving from their employers.
- Delivery personnel face severe exposure to the virus without paid sick leave, without work, and daily payouts.
- Recognising the key infrastructural role that delivery personnel are playing in the pandemic opens up a set of responsibilities for the government to take on.
A new reality?
- If governments around the world are ready to nationalise key infrastructure (like Spain’s private hospitals), can we use the food delivery workforce to deliver tests or essential medicines to those affected?
- With the Delhi government banning all dining-in and allowing only delivery, is the pandemic a time to think about a new form of regulation that brings private, socio-technological infrastructure into the public fold in times of crises, eventually to be released back into the forces of the market?
- Can the app service and app worker’s status as public infrastructure bolster a better interpretation of social protection that lasts beyond this crisis?
IRONING OUT WRINKLES IN INDIA’S PANDEMIC RESPONSE
Focus: GS-III Disaster Management
Why in news?
- Some critical weaknesses in the country’s health system can come in the way of a credible strategy to combat COVID-19
- Much concern about the novel coronavirus in India is understandably about the number of cases and related deaths.
Details
- It is important to remember that the vast majority (80%) of COVID-19 cases will be mild.
- The estimated mortality rate varies considerably between 3% to 0.25% of cases, and is much higher among the elderly.
- Notably, wealthier countries with stronger and better financed health systems such as Italy and China have struggled with containing COVID-19.
- As such, it is prudent to understand how well India’s health system can respond to COVID-19, especially since it is unclear how long this disease will persist. We believe that there are some critical weaknesses in India’s health system that can prevent a credible response to COVID-19.
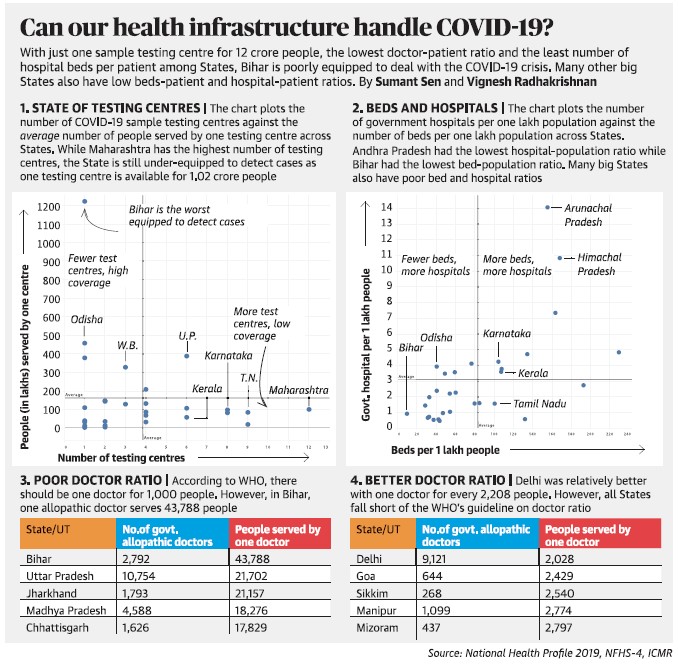
Ramping up hospital capacity
- Addressing the scarcity of hospital and intensive care unit (ICU) beds in India is critical for providing clinical support to severe COVID-19 cases.
- Without flattening India’s COVID-19 epidemic curve, our current hospital capacity is so low that it will be quickly overwhelmed if infections surge.
- It is critically important that India puts in place a strategy to ramp up hospital and ICU capacity, as well as provision for essential equipment such as ventilators and personal protective equipment for health workers. In both China and Italy, hospitals were rapidly constructed to accommodate infected patients.
- Tapping the resources in the private sector is particularly important. India’s health system is highly privatised and most of the country’s health-care capacity in terms of human resources, hospital beds, laboratories, and diagnostic centres is in the private sector.
- Recognising this, several State governments have initiated action, such as enlisting private laboratories for testing and using the private hospital bed capacity to treat positive patients.
- More of this is needed, as well as, engaging private hospitals in planning and coordinating the COVID-19 response.
Health workers are crucial
- Health-care workers are a critical resource for the COVID-19 response. They go into communities to carry out preventive care, trace potentially exposed people, and treat the infected.
- Primary-care providers, whether they are formally trained (e.g. medical officers, nurses, auxiliary nurse and midwives, pharmacists), or lay workers (accredited social health activists) or informal workers (rural (not registered) medical practitioners, or RMPs, drug shops) will likely be the first contact health workers for COVID-19 patients.
- Health workers also take on a disproportionate share of infections. Health worker safety is particularly important for India because it already faces a shortage of doctors and nurses.
Conclusion
- India like other countries faces important health system challenges in mounting a credible response to COVID-19. Many of these issues are not new.
- Addressing these health system issues will require much effort, financing, and, in some cases, not even entirely possible to remedy in the near future.
- How India deals with these health system issues in the days to come will make all the difference.
DEATH BY FIREWORKS: VIOLATIONS OF RULES IN HAZARDOUS INDUSTRIES
Focus: GS-III Industry and Infrastructure
Why in news?
- Unsafe working conditions and improper handling of inflammable raw materials continue to endanger lives in the fireworks industry.
- Tragedies have not been confined to Sivakasi, deemed the fireworks capital of the world, where most such units are concentrated.
- In and around Sivakasi, the manufacturing of firecrackers in makeshift unlicensed units, rough handling of chemicals by untrained and unskilled workers, spillage or overloading of chemicals during the filling process, and working outside permitted areas have been identified as major causes for past accidents.
Hazardous Industry
- Occasional accidents in an industry dealing in explosive materials may seem inevitable.
- But the probability of such mishaps can certainly be reduced by adopting safe work practices, complying with rules and through cohesive monitoring by Central and State licensing and enforcement authorities.
- Crackdowns against violators have been few and far between despite illegal sub-leasing of works to unlicensed cottage units becoming a widely acknowledged practice in the industry.
Way Forward
- The Chaitanya Prasad Committee, which examined, among other things, statutory and administrative shortcomings that led to the death of 40 workers at Om Shakti Fireworks Industries in 2012, noted the “conspicuous absence” of proper inspection mechanisms at various government departments.
- The committee recommended making sub-leasing of works by licensed units a cognisable penal offence; mandated inter-safety distances between sheds covered with earthen mounds; and provision of a smoothened pathway with a width of 1.5 metres, as part of industrial safety measures.
- Ground reports suggest these recommendations continue to be ignored, with sub-leasing of works still rampant.
- Regulators understandably complain of a lack of manpower in checking violations.
- The number of players has exponentially grown since the 1980s with 1,070 licensed units employing an estimated 10 lakh workers now. But safety is non-negotiable.
- The governments must walk the extra mile to enforce rules in a hazardous industry and prosecute violators.
- The industry too must self-regulate in its own interest.



