Why in news?
- The Indian Council of Medical Research (ICMR) on 9th April said the country has maintained a steady rate of COVID-19 positive cases — 3% to 5% over the past month and half — and has registered no significant increase in this trend so far.
- Kerala has won ICMR approval for the clinical protocol exploring the feasibility of an experimental therapy, convalescent plasma transfusion, which may be administered to severe COVID-19 patients.
- The Health Ministry added that two of its labs have started whole genome sequencing of the COVID-19 virus.
- Twenty companies are manufacturing PPE in India and orders for 1.7 crore PPE and 49,000 ventilators have already been placed, hence, there is no reason to believe any rumours or have any fear regarding PPE.
Details
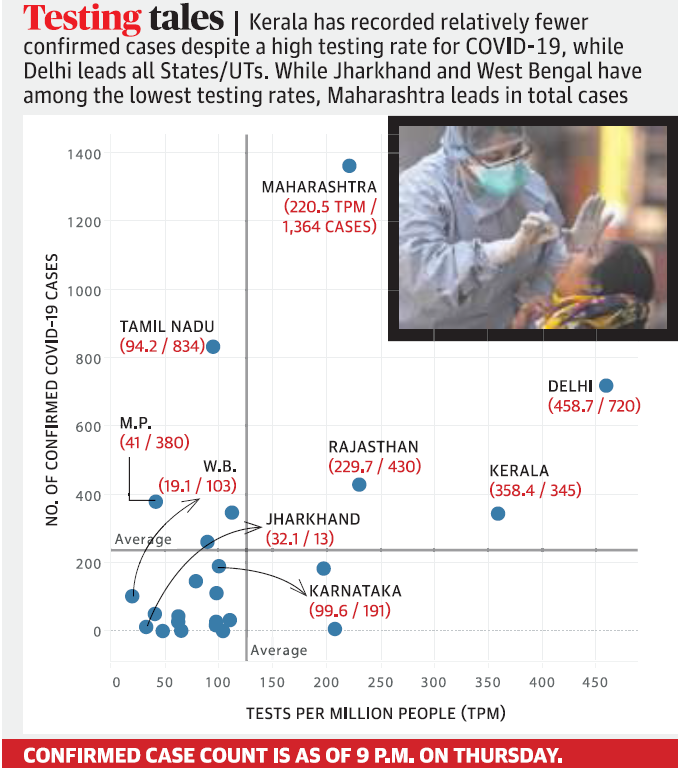
- The country currently has 5,865 confirmed cases and 169 deaths. This includes 591 new cases and 20 deaths in the last 24 hours.
- Of the 1,30,792 individuals tested as on April 9 – 5,734 samples tested positive. Positivity rate ranges between 3%-5% with no major increase.
- ICMR is in the final stages of drawing up a protocol for trial on convalescent plasma therapy, for which approvals have to be taken.
- 10 teams of COVID-19 specialists have been sent to nine States including — Bihar, Rajasthan, Uttar Pradesh, Tamil Nadu and Gujarat — to oversee preparedness and ensure maximum containment of cases.
- Indian Railways has produced about six lakh reusable face masks and over 4,000 litres of hand sanitiser; it has also converted 3,250 coaches into COVID-19 isolation units with beds. Total 5,000 coaches are to be converted
Current stand on Convalescent Plasma therapy
- At present there are no specific antiviral agents which have been found to be effective in the treatment of COVID-19.
- Convalescent plasma therapy is not new and have been used by doctors to treat critically ill patients during earlier epidemics too, during H1N1, SARS and Ebola.
- The proposal submitted to the Indian Council of Medical Research (ICMR) says that the Transfusion Medicine Department of the Sree Chitra Tirunal Institute for Medical Sciences and Technology would help the Health Department in exploring the possibility of convalescent plasma therapy for COVID-19 treatment.
- Getting the clinical protocol for administering plasma therapy cleared ahead by the ICMR is just part of Kerala’s preparedness, that should Kerala get severe COVID-19 patients during the second or third wave, they should be able to make use of this option.
- Drugs Controller General’s approval and institutional ethics committee approval would have to be there before the treatment can be administered.
- CP therapy shows a potential therapeutic effect and low risk in the treatment of severe COVID-19 patients.
- It said that one dose (200 ml) of convalescent plasma with a high concentration of neutralising antibodies was well-tolerated by patients and that it can rapidly reduce the viral load in patients and improve clinical symptoms significantly.
Plasma therapy
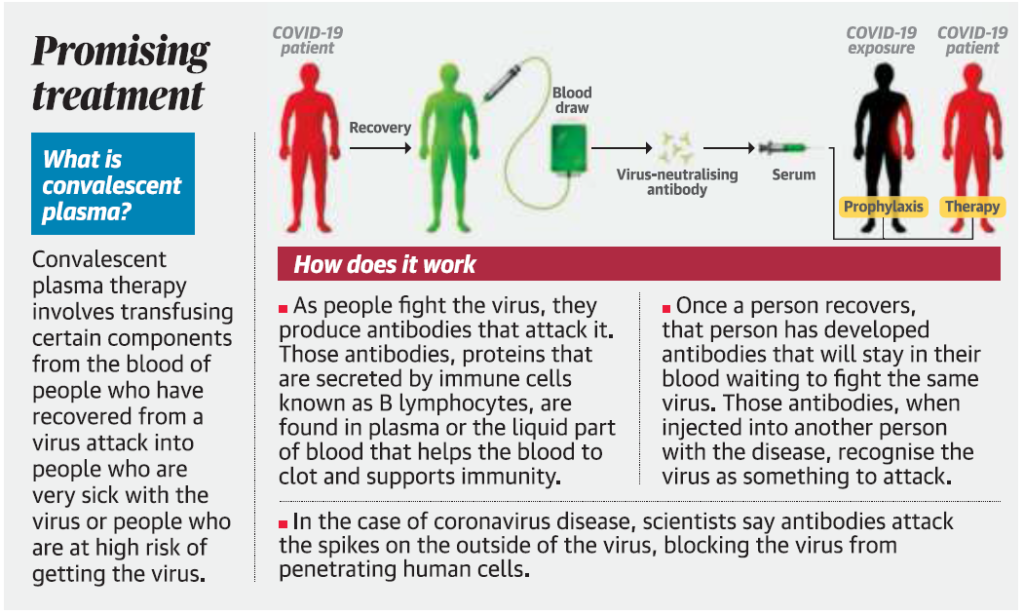
- Convalescent plasma (CP) therapy, a classic adaptive immunotherapy, has been applied to the prevention and treatment of many infectious diseases for more than one century.
- Over the past two decades, CP therapy was successfully used in the treatment of SARS, MERS, and 2009 H1N1 pandemic with satisfactory efficacy and safety
- Nevertheless, the potential clinical benefit and risk of convalescent blood products in COVID-19 remains uncertain.
- In this therapy, plasma from a COVID-19 recovered patient is transfused into an infected severely ill patient so that the specific antibodies in the blood of the recovered patient can help fight the infection.
- This is done for very ill patients on ventilators and has given good results in some studies abroad
Complications of plasmapheresis therapy
- Insertion of a rather large intravenous catheter can lead to bleeding, lung puncture (depending on the site of catheter insertion), and, if the catheter is left in too long, it can get infected.
- Bleeding or hematoma from needle placement
- Hypotension
- Potential exposure to blood products, with risk of transfusion reactions or transfusion transmitted diseases
- Suppression of the patient’s immune system
Odisha becomes first State to extend lockdown till April 30
- Odisha government on 9th April decided to extend lockdown until April 30 to contain the spread of COVID-19, Chief Minister Naveen Patnaik announced after the State Cabinet held a meeting through video-conferencing.
- Union government was also requested not to start airline and railway services till April 30.
- Educational institutions in Odisha would remain closed till June 17.
- Activities related to agriculture, animal husbandry and MGNREGS would be facilitated during the lockdown period adhering to social distancing norms.
- Food security of the people was a major priority of his government.



