Contents
- Kochi – Mangalore Natural Gas Pipeline
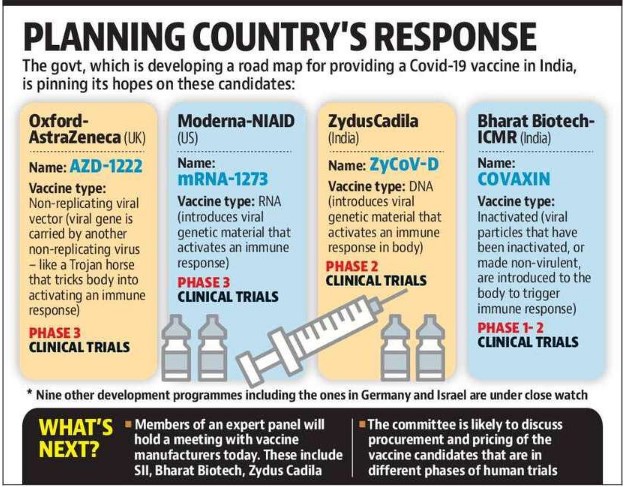
- Drugs Controller General of India (DCGI)
- Indigenously developed a DNA vaccine candidate against COVID-19
- Rashtriya Vayoshri Yojana (RVY) and Divyangjans
KOCHI-MANGALORE NATURAL GAS PIPELINE
Focus: GS 3;Infrastructure: Energy, Ports, Roads, Airports, Railways etc.
Why in News?
PM to dedicate Kochi – Mangalore Natural Gas Pipeline to the Nation on 5 January
About Background of Kochi-Mangalore Natural Gas Pipeline
- Prime Minister Shri Narendra Modi will dedicate the Kochi – Mangalore Natural Gas Pipeline to the nation on 5th January 2021
- The event marks an important milestone towards the creation of ‘One Nation One Gas Grid’.
- Governors and Chief Ministers of Karnataka and Kerala, along with Union Minister for Petroleum and Natural Gas will also be present on the occasion.
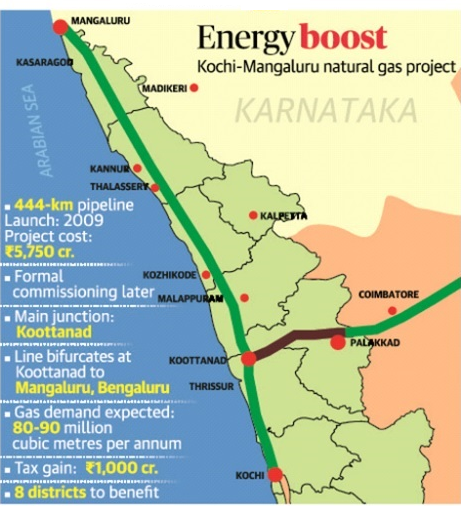
About Kochi-Mangalore Natural Gas Pipeline
- The 450 km long pipeline has been built by GAIL (India) Ltd.
- It has transportation capacity of 12 Million Metric Standard Cubic Metres per day, and will carry natural gas from the Liquefied Natural Gas (LNG) Regasification Terminal at Kochi (Kerala) to Mangalore (Dakshina Kannada district, Karnataka), while passing through Ernakulam, Thrissur, Palakkad, Malappuram, Kozhikode, Kannur and Kasaragod districts.
- The total cost of the project was about Rs. 3000 crore and its construction created over 12 lakh man-days of employment.
- Laying of the pipeline was an engineering challenge as the route of the pipeline necessitated it to cross water bodies at more than 100 locations.
- This was done through a special technique called Horizontal Directional Drilling method.
- The pipeline will supply environment friendly and affordable fuel in the form of Piped Natural Gas (PNG) to households and Compressed Natural Gas (CNG) to the transportation sector.
- It will also supply Natural Gas to commercial and industrial units across the districts along the pipeline.
- Consumption of cleaner fuel will help in improving air quality by curbing air pollution.
About Natural Gas and India’s Target
- Natural gas, is a naturally occurring hydrocarbon gas mixture consisting primarily of methane, but commonly including varying amounts of other higher alkanes, and sometimes a small percentage of carbon dioxide, nitrogen, hydrogen sulfide, or helium.
- Natural gas is a non-renewable hydrocarbon used as a source of energy for heating, cooking, and electricity generation.
- It is also used as a fuel for vehicles and as a chemical feedstock in the manufacture of plastics and other commercially important organic chemicals.
- Water, ethane, butane, propane, pentanes, hydrogen sulphide, carbon dioxide, water vapor, and occasionally helium and nitrogen may be present in a natural gas well.
- In order to be used for energy, the methane is processed and separated from the other components.
- According to the International Energy Agency (IEA), India is the world’s third largest consumer of oil.
- While the world average share of natural gas in the energy basket is 23%, it is only 6% for India.
- However, the government of India has set the target to make it 15% by 2030.
DRUGS CONTROLLER GENERAL OF INDIA (DGCI)
Focus: GS 2;Issues relating to development and management of Social Sector/Services relating to Health, Education, Human Resources.
Why in News?
Press Statement by the Drugs Controller General of India (DCGI) on Restricted Emergency approval of COVID-19 virus vaccine
Union Home Minister Shri Amit Shah hailed the approval granted today by the Drugs Controller General of India (DCGI) to the COVID-19 vaccines of the Serum Institute of India (SII) and Bharat Biotech
About Drugs Controller General of India (DGCI)
- Drugs Controller General of India is the head of department of the Central Drugs Standard Control Organization of the Government of India responsible for approval of licenses of specified categories of drugs such as blood and blood products, IV fluids, vaccines, and sera in India.
- Drugs Controller General of India, comes under the Ministry of Health & Family Welfare.
- DCGI also sets standards for manufacturing, sales, import, and distribution of drugs in India.
- DCGI heads the Indian drug regulatory body the Central Drugs Standard Control Organisation (CDSCO), whose functions include ensuring the quality of drugs and cosmetics sold in the country, approval of new drugs and regulating clinical trials.
- The central government have established 6 zonal offices of CDSCO (Central Drugs Standard Control Organisation) at Mumbai, Kolkata, Chennai, and Ghaziabad, Hyderabad, Ahmedabad, various Sub-Zonal offices and Port offices, which works in close collaboration with the state control administration and assist them in securing uniform enforcement of the Drug Act.

Functions of DGCI
- DCGI lays down the standard and quality of manufacturing, selling, import and distribution of drugs in India.
- Acting as appellate authority in case of any dispute regarding the quality of drugs.
- Preparation and maintenance of national reference standard.
- To bring about the uniformity in the enforcement of the Drugs and Cosmetics Act.
- Training of Drug Analysts deputed by State Drug Control Laboratories and other Institutions Analysis of Cosmetics received as survey samples from CDSCO (central drug standard control organisation)
- With the notification of Medical Device Rules 2017 by the Government of India, DCGI will also act as Central Licensing Authority (CLA) for the medical devices which fall under the purview of these rules.
- Out of four classes of medical devices from Class A to Class D, DCGI will be the direct licensing authority for Class C and Class D devices, whereas it will coordinate licensing for Class A and B devices through State drug controllers, who will act as State Licensing Authority or SLA.
INDIGENOUSLY DEVELOPED DNA VACCINE CANDIDATE AGAINST COVID-19
Focus: GS 2;Issues relating to development and management of Social Sector/Services relating to Health, Education, Human Resources.
Why in News?
DBT-BIRAC supported indigenously developed DNA Vaccine Candidate by Zydus Cadila, approved for Phase III clinical trials
About Indigenously Developed DNA Vaccine candidate Against COVID-19
- The Nation’s first indigenously developed DNA vaccine candidate against COVID-19, ZyCoV-D, by M/s Zydus Cadila has been approved by Drugs Controller General of India (DCGI), for conduct of the Phase III clinical trials.
- The establishment of the Nation’s first DNA vaccine platform is an important milestone for Atma Nirbhar Bharat and a big leap forward for Indian scientific research.
- The candidate has been supported by the National Biopharma Mission (NBM) under the aegis of BIRAC and the Department of Biotechnology, Government of India.
- Zydus Cadila completed Phase-I/II clinical trials of this DNA Vaccine candidate, in India, in more than 1,000 participants and interim data indicated that the vaccine is safe and immunogenic when three doses were administered intradermally.
- Based on the recommendations of the Subject Expert Committee, which reviewed the interim data, the DCGI has accorded permission for conducting Phase-III clinical trial in 26,000 Indian participants.
- The Department of Biotechnology, Government of India has partnered with Zydus Cadila, to address the need for accelerated development of an indigenous vaccine for COVID-19.
- The partnership exemplifies that such research endeavors demonstrate the Government’s focus on creating an ecosystem that nurtures and encourages new product innovation with societal relevance.
About DNA Vaccines
- A DNA vaccine is a type of vaccine that transfects a specific antigen DNA-coding sequence onto the cells of an immunized species.
- DNA vaccines are third generation vaccines, made up of small, circular pieces of bacterial DNA, plasmids. These plasmids are genetically engineered to produce specific antigens from a pathogen.
- DNA vaccination is a technique for protecting an animal against disease by injecting it with genetically engineered DNA so cells directly produce an antigen, resulting in a protective immunological response.
- DNA vaccine has a theoretical advantage in comparison to the other conventional vaccines
About Advantages of DNA Vaccine
- The immune response focused on the antigen of interest
- Cost-effective
- Less risk for infection
- Antigen presentation by MHC class I and class II molecules
- Long-term persistence of immunogen
About Limitations of DNA Vaccine
- Risk of affecting genes controlling cell growth
- Possibility of tolerance to the antigen
- Potential for atypical processing of bacterial and parasite proteins
- Limited to protein immunogens
RASHTRIYA VAYOSHRI YOJANA (RVY) AND DIVYANGJANS
Focus: GS 2;Welfare schemes for vulnerable sections of the population by the Centre and States and the performance of these schemes; mechanisms, laws, institutions and Bodies constituted for the protection and betterment of these vulnerable sections.
Why in News?
M/o Social Justice & Empowerment organizes Distribution camp to provide Aid & Assistive Devices to 1343 Senior Citizens and 564 Divyangjan in Bhatinda, Punjab
About Rashtriya Vayoshri Yojana (RVY) and Divyangjans
- The RVY was launched in 2017 at Nellore, Andhra Pradesh.
- Rashtriya Vayoshri Yojana (RVY) is a scheme for providing Physical Aids and Assisted-living Devices for Senior citizens belonging to BPL category.
- This is a Central Sector Scheme, fully funded by the Central Government.
- The expenditure for implementation of the scheme will be met from the “Senior Citizens’ Welfare Fund”.
- The Scheme will be implemented through the sole implementing agency – Artificial Limbs Manufacturing Corporation (ALIMCO), a PSU under the Ministry of Social Justice and Empowerment.
- Under the scheme, the physical aids will be provided only to the senior citizens of the nation.
- This implies those who are aged above 60 years will get free assisted living aids and physical devices which are required for their sustainability.
- Also the govt. has selected the list of cities where the scheme will be implemented.
- The main criteria for the senior citizens to get full benefits of Rashtriya Vayoshri Yojana (RVY) scheme is that they must belong to BPL family and should hold valid BPL card issued by the concerned authority.
- Beneficiaries in each district will be identified by the State Governments/UT Administrations through a Committee chaired by the Deputy Commissioner/District Collector.
- As far as possible, 30% of the beneficiaries in each district shall be women.

About Significance of RVY Scheme
- According to Census 2011, there are 10.38 crores of senior citizens.
- And of these, more than 70% of them live in rural areas.
- Therefore, it is essential to take special care of them through these camps organized under Rashtriya Vayoshri Yojana.



