Focus: GS-III Science and Technology
Why in news?

- The Indian Council of Medical Research (ICMR) has tied up with Bharat Biotech India (BBIL) to “fast-track” trials of the Hyderabad firm’s Covid-19 vaccine, Covaxin.
- ICMR chief wrote to all the trial sites stating that all of the clinical trials had to be completed by August 15 in time for a “public launch” of the vaccine.
- Even vaccine candidates like those of AstraZeneca and the University of Oxford, at the most advanced stages of trials globally, do not expect to launch their product before the end of the year.
What is Covaxin?
Covaxin is BBIL’s vaccine candidate to immunise people against Covid-19.
- The vaccine candidate was developed in collaboration with ICMR’s National Institute of Virology (NIV).
- Once the vaccine is injected into a human, it has no potential to infect or replicate, since it is a killed virus. It just serves to the immune system as a dead virus and mounts an antibody response towards the virus.
What stage of testing is Covaxin at right now?
- The Drug Controller General of India, who heads apex drug regulatory body Central Drugs Standard Control Organisation (CDSCO), has given BBIL approvals for phase I and II clinical trials so far.
- According to the registry, BBIL has estimated the trials to take one year and three months. Of this, phase I trials alone will take at least a month.
- The ultimate goal is the selection of a safe, well-tolerated, and immunogenic intramuscular vaccine, which will be further evaluated in the phase 2 study.
Is this possible?
- Most experts are sceptical of the possibility of fast-tracking trials to such an extent that all three phases can be concluded within a month and a half.
- Even the most ambitious companies in the world that are in more advanced stages of development for their Covid-19 vaccines have a longer time-frame.
What other Covid-19 vaccines are being tested in India and around the world?
- Currently, there are more than 100 vaccine candidates at different stages of trials across the world.
- The AstraZeneca-Oxford vaccine has already progressed to the Phase-III trials stage, and this will, in the best-case scenario, only be available towards the end of 2020.
- Pfizer (in collaboration with Germany’s BioNTech) have already been giving patients its experimental Covid-19 vaccine at low- and mid-level doses.
What are Clinical trials?
- Clinical trials or Human Trials are experimental studies on human participants that helps to determine the safety concerns, effectivity and potency of a particular treatment (drugs, vaccines, dietary supplements, dietary choices, medical devices, etc.)
- While preclinical research answers basic questions about a drug’s safety, it is not a substitute for studies of ways the drug will interact with the human body – Hence, Clinical Trials are very important in understanding the effects of administering a particular treatment.
Human Clinical Trial Phases
Preclinical Phase (Phase 0)
- Drug and device testing begin with extensive laboratory research which can involve years of experiments in animals and human cells.
- If the initial laboratory research is successful, researches send the data to the Food and Drug Administration (FDA) for approval to continue research and testing in humans.
- Once approved, human testing of experimental drugs and devices can begin and is typically conducted in four phases.
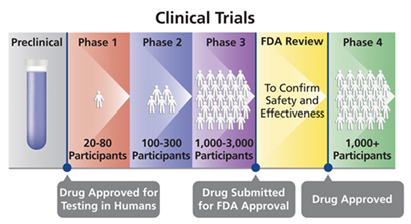
Phase I
- Phase I studies assess the safety of a drug or device. Usually includes a small number of healthy volunteers (20 to 100).
- The study is designed to determine the effects of the drug or device on humans including how it is absorbed, metabolized, and excreted.
- This phase also investigates the side effects that occur as dosage levels are increased.
Phase II
- Phase II studies test the efficacy of a drug or device. Involves up to several hundred patients.
- Most phase II studies are randomized trials where one group of patients receives the experimental drug, while a second “control” group receives a standard treatment or placebo.
- Often these studies are “blinded” which means that neither the patients nor the researchers know who has received the experimental drug.
Phase III
- Phase III studies involve randomized and blind testing in several hundred to several thousand patients. This large-scale testing, which can last several years.
- Once Phase III is complete, a pharmaceutical company can request FDA approval for marketing the drug.
Phase IV
- Phase IV studies, often called Post Marketing Surveillance Trials, are conducted after a drug or device has been approved for consumer sale.
- Phase IV studies can result in a drug or device being taken off the market or restrictions of use could be placed.
-Source: Indian Express



