The Israeli Defence Minister earlier this week said that scientists at the country’s main biological research institute have made a “significant breakthrough” in developing an antibody to the novel coronavirus, as the researchers wrapped up the development phase and moved to patent and mass produce the potential treatment. A statement said that the antibody’s development had been completed and that the institute was in the process of patenting the find “and in the next stage, researchers will approach international companies to produce the antibody on a commercial scale”.
On this edition of The Big Picture participants analyse the antibody breakthrough against coronavirus and other treatments.
What is this the antibody breakthrough?
This breakthrough is actually in finding a set of antibodies or we can say free antibodies that can neutralize the virus and blocking from infecting the human cells. Antibodies meet three important parameters.
First, they are fully human monoclonal;
Second, they are meant to bind two distinct parts of the corona virus and
Third, in that sense block it and not enabling it to infect the human cell
Institutes of biological Research demonstrated the ability of those antibodies to neutralize the corona Virus. these three parameters together are a breakthrough which will later enable us to achieve a full treatment in much less time that we needed. we will dissipate through it shortens the time for full treatment dramatically
What is antibody treatment and how is it different from a vaccine?
Antibody drugs are powerful tools of modern medicine, capable of specifically binding and disabling all sorts of targets, but they are complicated and expensive to develop.
Vaccines let your body do the hard work and develop specialized antibodies to fight infections for itself. In a sense, all the research done to create antibody drugs is just trying to replicate in a lab what the human body does on its own!
How does the antibody treatment really work?
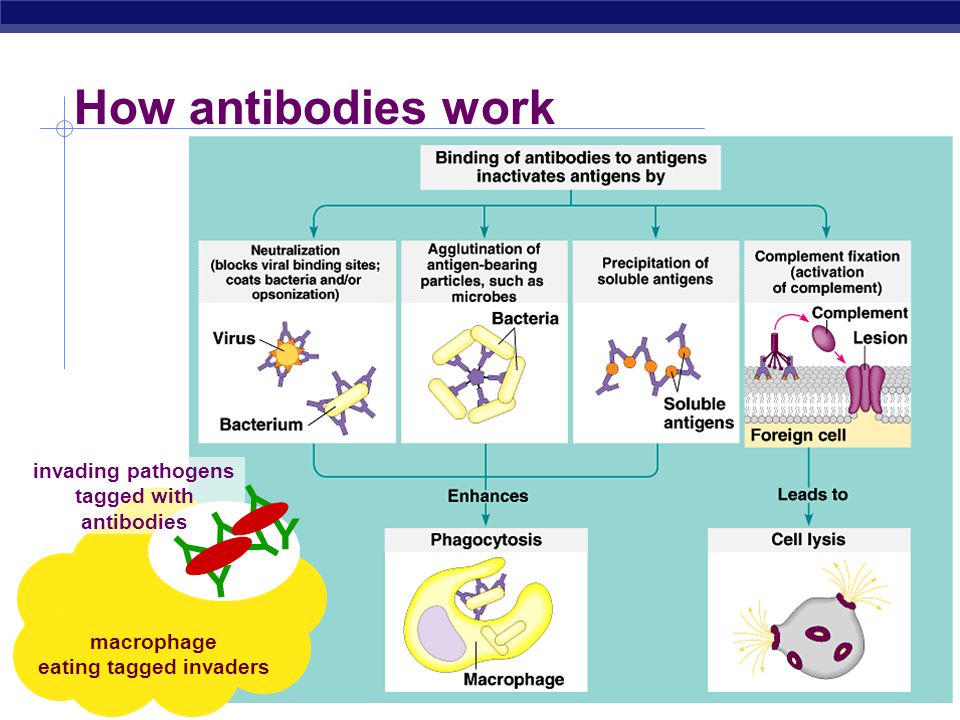
Viruses can also be removed from the body by antibodies before they get the chance to infect a cell. Antibodies are proteins that specifically recognise invading pathogens and bind (stick) to them. This binding serves many purposes in the eradication of the virus:
- Firstly, the antibodies neutralise the virus, meaning that it is no longer capable of infecting the host cell.
- Secondly, many antibodies can work together, causing virus particles to stick together in a process called agglutination. Agglutinated viruses make an easier target for immune cells than single viral particles.
- A third mechanism used by antibodies to eradicate viruses, is the activation of phagocytes. A virus-bound antibody binds to receptors, called Fc receptors, on the surface of phagocytic cells and triggers a mechanism known as phagocytosis, by which the cell engulfs and destroys the virus.
- Finally, antibodies can also activate the complement system, which opsonises and promotes phagocytosis of viruses. Complement can also damage the envelope (phospholipid bilayer) that is present on some types of virus
What is monoclonal antibodies?
Monoclonal means “coming from one clone of antibody producing cells” these are called plasma cells. These antibodies are all the same and are all coming from the same clone of cells. And should all bind to the same antigen and binding site.
Polyclonal antibodies are a mixture of antibodies from several clones of plasma cells. And should bind to more antigens or different binding sites on one kind of antigen.

The real breakthrough is that – now cheaper mass-producing technologies are available and that really can make this affordable in contrast to vaccines which will require greater development phase, trials and then of course making this available for the vulnerable population or ideally an entire population. The advantage here is that it will be therapeutic in a relatively early phase of the disease so the patient will be both protected as well as rendered non-infectious.
What are the research taking place around the world to develop a vaccine or treatment?
United States:
Gilead Sciences has developed Remdesivir, a broad-spectrum antiviral medication that is being tested as a specific treatment for COVID-19 infections. The drug has been authorised for emergency use in USA .
Another pharmaceutical company, Regeneron stated that its ‘anti-body’ treatment drug could also be available by September.
China:
Beijing-based Sinovac Biotech is in discussion with regulators in other countries, and the World Health Organization, to launch phase III clinical trials of the vaccine in regions where the novel coronavirus is still spreading rapidly.
Chinese scientists have three other potential COVID-19 vaccines in human trials: one from the Chinese military in collaboration with Tianjin-based CanSino Biologics Inc., and two from state-owned China National Biotec Group.
Italy:
Luigi Aurisicchio, CEO of Takis, the firm developing the medication, stated that for the first time a coronavirus candidate vaccine developed by them was able to neutralise the virus in human cells.
A researcher from Takis stated “as far as we know, we are the first in the world so far to demonstrate neutralisation of coronavirus by a vaccine. We expect this to happen in humans too.”
Israel:
Defence Minister of Israel, Naftali Bennett declared that the Israel Institute for Biological Research (IIBR) has developed an antibody to neutralise the COVID-19 virus. It can attack the virus within the bodies of the infected and neutralise it.
India:
More than 30 vaccines are in various stages of development in India, scientists informed Prime Minister Narendra Modi on May 5.
University of Oxford’s vaccine is also being developed with multiple partners including India’s Serum Institute of India. The WHO document says out of the 100 projects under pre-clinical stages, Indian companies like Zydus Cadila, Codagenix-Serum Institute of India, Indian Immunologicals with Griffith University, Bharat Biotech in association with Thomas Jefferson University, Biological E Ltd, and the UW Madison-FluGen-Bharat Biotech combine are all working on potential vaccine candidates to cure the coronavirus infection.
United Kingdom:
Scientists at the Jenner Institute of Oxford University have stated to have made a potential vaccine for coronavirus. The vaccine is being developed with multiple partners including the Serum Institute of India.
Interferon Beta, a drug developed by biotech company Synairgen has been injected in patients as part of initial trials.
How long does it usually take in treatment like this to become commercially available and what are the various phases of testing and the various phases that people’s like this has to go through?
Regulatory processes are now being expedited all across the world by all regulatory agencies so making of antibodies would actually require less time than then approving or licensing a vaccine. A lot of in vitro tests can actually happen which means in the laboratory and that probably is the promise that the Israeli Institute is working on it.
World is racing to find multiple initiatives and vaccines. This is not one magic bullet in isolation but it is really the battery of interventions that we would potentially be looking at in the horizon.
What India has been doing as well what kind of treatment has India tried?
ICMR approved the anti malaria drug hydroxychloroquine for as a prophylactic use in risk groups as well as in healthcare workers.
India is also part of the WHO solidarity trial and India has identified four hospitals as of clinical trial sites and others are in the process of being identified.
India is looking at convalescent plasma therapy.
India also enrolled patients for randomized controlled trial where there are two sets of patient groups, basically one which receives the treatment and one which does not receive the treatment so it establishes the whole thing in a much strong way .
ICMR has approved given out guidelines for use of HIV combination drug Lupin that can be used in moderately severe patients and hospitals will have to monitor those patients very carefully and with the consent but these are all supportive treatments
What are the cooperation taking place between India and Israel?
Israel has suggested 50 topics in health and science where it can cooperate with India to find solutions to the Covid-19 crisis even as it works to bring together private enterprises from both sides to develop technologies to manage and overcome the pandemic.
Israel is working to put together start-ups and entrepreneurs from both sides to work on finding solutions to different aspects of the Covid-19 crisis. The focus is not just on equipment to prevent infections and defeat the virus but also on facilitating new ways of doing things amid the unprecedented lockdown.
Israel is also keen to share data on Covid-19 with India as having more data to analyse will help uncover patterns and characteristics of the disease
Background: What is WHO-led Solidarity Trial?
“Solidarity” is an international clinical trial to help find an effective treatment for COVID-19, launched by the World Health Organization and partners.
The Solidarity Trial will compare four treatment options against standard of care, to assess their relative effectiveness against COVID-19. By enrolling patients in multiple countries, the Solidarity Trial aims to rapidly discover whether any of the drugs slow disease progression or improve survival. Other drugs can be added based on emerging evidence.
Based on evidence from laboratory, animal and clinical studies, the following treatment options were selected:
- Remdesivir; (previously tested as an Ebola treatment)
- Lopinavir/Ritonavir; (For HIV)
- Lopinavir/Ritonavir with Interferon beta-1a; and (used to treat multiple sclerosis)
- Chloroquine or Hydroxychloroquine. (used to treat malaria and rheumatology conditions respectively)



