Contents
- ICMR and Launch of ‘Covaxin’
- From Pinaka to Astra: DAC approved new weapons
- India in UNSC: In an uncertain world
ICMR AND LAUNCH OF ‘COVAXIN’
Focus: GS-III Science and Technology
Why in news?

- The Indian Council of Medical Research (ICMR) has tied up with Bharat Biotech India (BBIL) to “fast-track” trials of the Hyderabad firm’s Covid-19 vaccine, Covaxin.
- ICMR chief wrote to all the trial sites stating that all of the clinical trials had to be completed by August 15 in time for a “public launch” of the vaccine.
- Even vaccine candidates like those of AstraZeneca and the University of Oxford, at the most advanced stages of trials globally, do not expect to launch their product before the end of the year.
What is Covaxin?
Covaxin is BBIL’s vaccine candidate to immunise people against Covid-19.
- The vaccine candidate was developed in collaboration with ICMR’s National Institute of Virology (NIV).
- Once the vaccine is injected into a human, it has no potential to infect or replicate, since it is a killed virus. It just serves to the immune system as a dead virus and mounts an antibody response towards the virus.
What stage of testing is Covaxin at right now?
- The Drug Controller General of India, who heads apex drug regulatory body Central Drugs Standard Control Organisation (CDSCO), has given BBIL approvals for phase I and II clinical trials so far.
- According to the registry, BBIL has estimated the trials to take one year and three months. Of this, phase I trials alone will take at least a month.
- The ultimate goal is the selection of a safe, well-tolerated, and immunogenic intramuscular vaccine, which will be further evaluated in the phase 2 study.
Is this possible?
- Most experts are sceptical of the possibility of fast-tracking trials to such an extent that all three phases can be concluded within a month and a half.
- Even the most ambitious companies in the world that are in more advanced stages of development for their Covid-19 vaccines have a longer time-frame.
What other Covid-19 vaccines are being tested in India and around the world?
- Currently, there are more than 100 vaccine candidates at different stages of trials across the world.
- The AstraZeneca-Oxford vaccine has already progressed to the Phase-III trials stage, and this will, in the best-case scenario, only be available towards the end of 2020.
- Pfizer (in collaboration with Germany’s BioNTech) have already been giving patients its experimental Covid-19 vaccine at low- and mid-level doses.
What are Clinical trials?
- Clinical trials or Human Trials are experimental studies on human participants that helps to determine the safety concerns, effectivity and potency of a particular treatment (drugs, vaccines, dietary supplements, dietary choices, medical devices, etc.)
- While preclinical research answers basic questions about a drug’s safety, it is not a substitute for studies of ways the drug will interact with the human body – Hence, Clinical Trials are very important in understanding the effects of administering a particular treatment.
Human Clinical Trial Phases
Preclinical Phase (Phase 0)
- Drug and device testing begin with extensive laboratory research which can involve years of experiments in animals and human cells.
- If the initial laboratory research is successful, researches send the data to the Food and Drug Administration (FDA) for approval to continue research and testing in humans.
- Once approved, human testing of experimental drugs and devices can begin and is typically conducted in four phases.
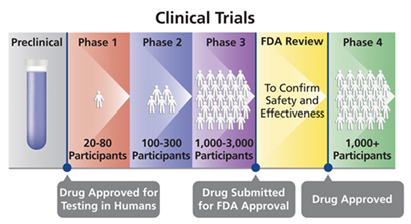
Phase I
- Phase I studies assess the safety of a drug or device. Usually includes a small number of healthy volunteers (20 to 100).
- The study is designed to determine the effects of the drug or device on humans including how it is absorbed, metabolized, and excreted.
- This phase also investigates the side effects that occur as dosage levels are increased.
Phase II
- Phase II studies test the efficacy of a drug or device. Involves up to several hundred patients.
- Most phase II studies are randomized trials where one group of patients receives the experimental drug, while a second “control” group receives a standard treatment or placebo.
- Often these studies are “blinded” which means that neither the patients nor the researchers know who has received the experimental drug.
Phase III
- Phase III studies involve randomized and blind testing in several hundred to several thousand patients. This large-scale testing, which can last several years.
- Once Phase III is complete, a pharmaceutical company can request FDA approval for marketing the drug.
Phase IV
- Phase IV studies, often called Post Marketing Surveillance Trials, are conducted after a drug or device has been approved for consumer sale.
- Phase IV studies can result in a drug or device being taken off the market or restrictions of use could be placed.
-Source: Indian Express
FROM PINAKA TO ASTRA: DAC APPROVED NEW WEAPONS
Focus: GS-III Science and Technology, Internal Security
Why in news?
- The Defence Acquisition Council (DAC) chaired by Defence Minister cleared several proposals worth close to Rs 39,000 crore that will boost the combat capabilities of all three services—Army, Navy and the Air Force.
- The most prominent of these proposals include missile systems for the three services, and additional fighter jets for the Air Force.
- The aircraft will be bought from Russia, and also from the domestic Hindustan Aeronautics Limited (HAL).
- The air-to-air Astra missile, Pinaka munitions and Land Attack Cruise Missile are state of the art systems developed by DRDO. Armed forces and industry will be immensely benefitted with the manufacturing of these indigenous systems.
Pinaka missile system for the Army
- Pinaka missile system is an all-weather, indirect fire, free flight artillery rocket system, according to the DRDO
- It provides a unique capability to accurately deliver a devastatingly lethal and responsive fire against a variety of area targets such as exposed enemy troops, armored and soft skin vehicles, communication centers, air terminal complexes, fuel and ammunition dumps.
Astra Missiles for Navy and Air Force
- Astra Missiles provide the Forces with Beyond Visual Range (BVR) capability.
- The BVR class of Air-to-Air Missile (AAM) system designed to be mounted on fighter aircraft, designed to engage and destroy highly manoeuvring supersonic aircraft.
- The ASTRA Mk-I Weapon System integrated with SU-30 Mk-I aircraft is being inducted into the Indian Air Force (IAF).
- It can be launched in autonomous and buddy mode of operation with features for Lock-On-Before Launch (LOBL) and Lock-On After Launch (LOAL).
Development of Long-Range Land Attack Cruise Missile Systems (LRLACM) for Navy and Air Force
- It is aimed at enhancing India’s firing range from between 400 km and 500 km of the Brahmos to 1000 km.
- The seeker development and testing by DRDO laboratories are demonstrated and is at a high-level of readiness.
-Source: Indian Express
INDIA IN UNSC: IN AN UNCERTAIN WORLD
Focus: GS-II International Relations
Introduction
India will be back in the United Nations Security Council (UNSC) for a two-year term beginning January 1, 2021 at a critical time in the history of the UN.
All about the contest
- The basic contest for the non-permanent seats takes place in the respective regional groups and their sub-groups.
- Voting in the General Assembly is to fulfil the requirement of countries having to secure a two-thirds majority of the member states.
- If there is regional endorsement, all countries, except those with any grievance against the candidates, vote for them and they sail through easily.
- But regional endorsement is becoming difficult as countries inscribe their names years in advance and those squatting countries have to be persuaded to vacate the place through various means.
- Last time, it was Kazakhstan which vacated the place for India; this time, it was Afghanistan.
What voting against a country might mean?
- When it comes to Voting in the General Assembly, usually no one gets all votes cast, as the ballot is secret and adversaries may vote against the candidates.
- Out of the 192 votes cast, India got 184 and no one will ever know the eight countries that did not vote for India.
- But it is a matter of concern that there are so many countries with grievances against India.
Permanent membership
- India’s election as a non-permanent member has understandably ignited the hope that its quest for permanent membership of the Council may succeed.
- A majority of the UN members are against the privileges of the permanent members, particularly the veto.
- The debate of increasing the number of Permanent Members of the UNSC has thrown up many ideas, but till today, none of the proposals has the possibility of securing two-thirds majority of the General Assembly and the votes of the five permanent members.
Significance of India’s Current Position as a non-permanent member
- India will have a higher profile at the UN for the next two years as the non-permanent members have a collective veto over every resolution in the Council.
- Permanent members can prevent adoption of resolutions by themselves, but they need at least nine votes to get a resolution passed.
- India will also have a rare peep into the consultations chamber of the UNSC, which is closed to non-members of the Council. It is there that hard negotiations take place without any public record, characterised by arm-twisting and threats of veto.
- The pressure of work of the mission will also increase because India will get involved in many issues in which it may not have any direct interest.
- Since India does not have a veto, it shall have to proceed cautiously not to offend anyone.
Click Here to read more about the UNSC and India’s Membership
-Source: The Hindu



