Why in news?
The Government of India announced on 7th March 2020 that it had rescinded its earlier ban on the export of malaria drug hydroxychloroquine (HCQ), which is now being used in countries such as the U.S. as a possible line of treatment for COVID-19, setting off a controversy over whether the two-day old blanket ban was overturned under pressure from Washington.

What the Ministry of External Affairs had to say?
- In view of the humanitarian aspects of the pandemic, it has been decided that India would licence paracetamol and HCQ in appropriate quantities to all our neighbouring countries who are dependent on our capabilities.
- We will also be supplying these essential drugs to some nations that have been particularly badly affected by the pandemic.
- The latest decision, which was taken at a high-level meeting on April 6, effectively overturns the previous notification.
- The MEA indicated that pre-existing orders, mainly to the U.S., Brazil and European countries would be fulfilled, depending on the stock position and domestic demand for the drug.
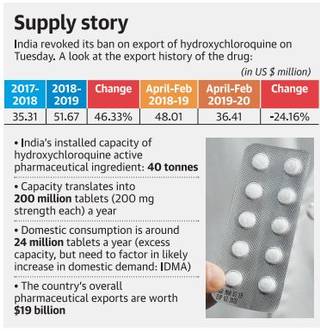
Background
- In a notification on March 25, the government placed HCQ on a restricted items list, and then put a blanket ban on any export of the drug on April 4.
- The MEA announcement came hours after U.S. President Donald Trump said that India could invite “retaliation” if it withheld supplies of HCQ for which the U.S., Brazil and other countries have already placed advance orders.
- “I spoke to [PM Modi] yesterday, a good talk. I would be surprised [if India refused to supply HCQ], because India does very well by the U.S. For many years they have been taking advantage of the United States on trade….I spoke to him and said, we appreciate your allowing the supply to come out. If he doesn’t allow it come out, that would be ok, but of course there may be retaliation, why wouldn’t there be?” Mr. Trump said.
- While the drug’s efficacy is not yet clinically proven, Mr. Trump has been a proponent of its use.
Criticisms
- The Opposition criticised the Union government for the quick capitulation in the face of a veiled threat by United States President Donald Trump on the ban on export of anti-malarial drug hydroxychloroquine.
- The government should think deeply about the export of essential medicines as our citizens have the first right over them.
- While the humanitarian gesture made by the Indian government was welcome, it should have raised concern over the language used by Mr. Trump.
MEA’s Response to Criticisms
- MEA is in touch with the Department of Pharmaceuticals, which has assured us that we have enough supply.
- This drug is not recommended for use in general population. In India, it is still given as a trial drug to a restricted category of people, and should not be taken without doctor’s advice.
- The MEA also denied criticism that its decision to reverse the ban had been taken under pressure from the U.S. and other countries.
- Like any responsible government, our first obligation is to ensure that there are adequate stocks of medicines for the requirement of our own people.
- In order to ensure this, some temporary steps were taken to restrict exports of a number of pharmaceutical products.



