Context:
A new point-of-care test developed by ICMR-National Institute of Virology has been found to be highly sensitive in the rapid diagnosis of Kyasanur forest disease (KFD), also known as monkey fever, which is an emerging public health problem in the country.
Relevance:
Prelims, GS-III: Science and Technology, GS-II: Social Justice (Health related issues)
Dimensions of the Article:
- About the Kyasanur Forest disease
- About the new Point-of-care Test by ICMR-NIV
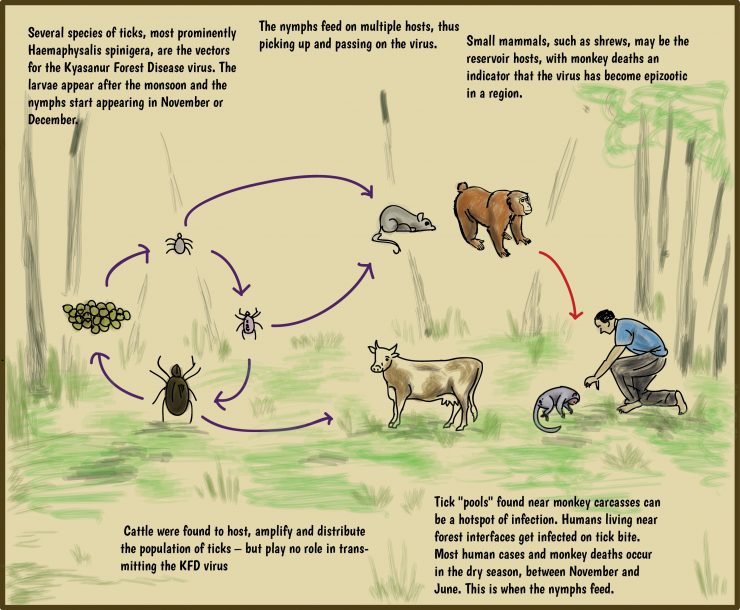
About the Kyasanur Forest disease
- Kyasanur forest disease (KFD) is a tick-borne viral haemorrhagic (accompanied by or produced by loss/escape of blood from a broken blood vessel, either inside or outside the body) fever ENDEMIC TO SOUTH-WESTERN PART OF INDIA.
- The disease is caused by a virus belonging to the family Flaviviridae.
- KFDV is transmitted to humans through the bite of infected hard ticks (Haemaphysalis spinigera) which act as a reservoir of KFDV.
- Patients may experience abnormally low blood pressure, and low platelet, red blood cell, and white blood cell count.
- A variety of animals are thought to be reservoir hosts for the disease, including porcupines, rats, squirrels, mice, and shrews.
- Monkeys are the main amplifying hosts for KFD virus and they are also sufferers, hence, the name Monkey Fever.
- Man is a terminal host and there no human-to-human transmission because the human domestic environment does not sustain the ticks.
- Prevention is by vaccination, as well as preventive measures such as protective clothing and tick population control.
About the new Point-of-care Test by ICMR-NIV
- A new point-of-care test, developed by Indian Council of Medical Research (ICMR)-National Institute of Virology, is made for diagnosis of Kyasanur Forest Disease (KFD).
- It would be beneficial for the diagnosis of KFD as the outbreaks mainly happen in remote areas, where there is lack of well-equipped sample handling and laboratory testing facilities.
- It would be useful in quick patient management and controlling further spread of the virus.
- It includes a battery-operated Polymerase Chain Reaction (PCR) analyser, which is a portable, lightweight and universal cartridge-based sample pre-treatment kit and nucleic acid extraction device that aid in sample processing at the point of care.
-Source: Indian Express



